BY MATTHEW GU
1. Introduction
The sun blazed mercilessly overhead, baking the dusty paths of a small village in
central Congo. Jeanne, a mother, shielded her eyes from the harsh glare as she
carefully descended the hill, balancing a basket of fruit on her head. Her youngest
daughter’s laughter echoed faintly from the village below.
But halfway down, Jeanne felt it: a peculiar tingling in her chest. At first, she thought
it might be the heat or exhaustion from the day’s work. The sensation grew rapidly,
spreading like wildfire through her body, searing muscle and bone. By the time the
fiery pain reached its peak, Jeanne dropped to her knees and collapsed into the
dust.
What Jeanne didn’t know in those agonising moments was that her body was
succumbing to one of the deadliest viruses in human history:
Ebola.
Jeanne was not alone. Since its discovery in 1976, the Ebola virus has ravaged
communities across West and Central Africa, claiming thousands of lives. Families
like hers have been shattered, entire villages emptied of laughter and light. Although
Ebola only claimed the lives of 12,000, its fatality rate has fluctuated between 25-
90%, an average of 50% depending on circumstances and responses making Ebola
one of the deadliest viruses in history (WHO).
Ebola, also known as Ebola Virus Disease (EVD), is a severe viral haemorrhagic
fever affecting humans and other primates, caused by ebolaviruses. Symptoms
typically appear between two days and three weeks after exposure (Modrow &
Falke, 2013). The disease was first identified in 1976 during simultaneous outbreaks
in Nzara, South Sudan, and Yambuku, a village near the Ebola River in the DRC,
from which it derives its name (WHO).
Figure 1: Map of Africa and the Ebola River.
Between 1976 and 2012, there were 24 recorded relatively small Ebola outbreaks,
resulting in 2,387 cases and 1,590 deaths (Dixon & Schafer, 2014). The largest and
most devastating outbreak occurred in West Africa between December 2013 and
January 2016, with 28,646 cases and 11,323 deaths (CDC, 2014). On March 29,
2016, the epidemic was declared no longer an international emergency.
2. Ebola
Ebola begins with sudden flu-like symptoms, including fever, sore throat, muscle
pain, weakness, decreased appetite, and headaches (Goeijenbier & Kampen, 2014).
These initial symptoms are followed by nausea, vomiting, and diarrhoea alongside
impaired liver and kidney function leading to severe dehydration. In some cases,
patients may experience shortness of breath, chest pain, swelling, and confusion
(Magill, 2013). In about 50% of cases, patients develop a maculopapular rash. As the disease
progresses, internal and external bleeding may occur, with all infected individuals
exhibiting reduced blood clotting (Hoenen, 2006). Bleeding from mucous membranes
or needle puncture sites is observed in about 40–50% of cases (King, 2021). This
may be introduced as vomiting blood, coughing up blood, or blood in the stool.
Bleeding under the skin can cause petechiae, purpura, ecchymoses, or hematomas,
particularly at injection sites (Feldmann & Geisbert, 2011). In some cases, bleeding
into the whites of the eyes has been noted (Shantha, 2016). While heavy bleeding is
uncommon, gastrointestinal haemorrhaging occurred in approximately 58% of cases
during the 2001 Gabon outbreak (Cappell, 2015).
Ebola is transmitted through direct contact with infected bodily fluids, such as blood,
breast milk, or semen, or through contact with contaminated surfaces (WHO). The
virus will usually remain in bodily fluids for several months (CDC, 2015). Airborne
transmission among humans is theoretically possible due to the presence of Ebola
virus particles in saliva, which can be released into the air with a cough or sneeze,
but observational data from previous epidemics both in nature and in laboratories
suggest the actual risk of airborne transmission is low (Jones, 2015).
Survivors often have ongoing muscular and joint pain, liver inflammation, and
decreased hearing, and may have continued tiredness, weakness, decreased
appetite, and difficulty returning to pre-illness weight (Tosh, Sampathkumar, 2014).
Problems with vision may also develop (WHO).
3. Virology
Ebola virus, a member of the Filoviridae family, is classified as a filamentous,
enveloped, negative-sense single-stranded RNA virus. It exhibits a distinctive
filamentous appearance, with virions typically measuring between 800 nm and 1,400
nm in length and approximately 80 nm in diameter. Under electron microscopy, these
virions may appear as coiled U-shaped, circular, 6-shaped, or branched filaments
(Feldmann & Geisbert, 2011).
Figure 2: Image of the Ebola Virus.
The life cycle of EVD begins with the attachment of its virions to specific cell-surface
receptors such as C-type lectins, DC-SIGN, or integrins. These receptors facilitate
viral entry into the host cell, a process that is aided by the viral glycoprotein (GP1,2).
Following attachment, the viral envelope fuses with the cellular membrane, allowing
the virus to enter the cell (Misasi, 2014). Once inside, the virions are transported to
acidic compartments such as endosomes and lysosomes, where the glycoprotein is
severed. This cleavage enhances the virus’s ability to bind to internal cellular
proteins, enabling fusion with internal membranes and the release of the viral
nucleocapsid into the cytoplasm (Misasi, 2014).
The viral nucleocapsid contains the RNA genome and associated proteins essential
for replication. The Ebola virus’s RNA polymerase, encoded by the L gene, partially
uncoats the nucleocapsid and begins transcribing the viral genes into positive-strand
mRNAs. These mRNAs are translated into both structural and non-structural proteins
required for viral assembly. The nucleoprotein is the most abundantly produced viral
protein, and its concentration in the host cell dictates when the polymerase
transitions from transcription to genome replication (Olejnik, 2011).
Newly synthesised genomes and structural proteins self-assemble into viral particles
near the inner surface of the host cell membrane. The assembled virions then bud
off from the cell, acquiring their lipid envelope from the host membrane during this
process. These mature progeny particles are capable of infecting new cells,
therefore perpetuating the viral life cycle. However, studying this life cycle is
challenging due to the extreme virulence of the Ebola virus, which poses significant
biosafety risks (Feldmann & Geisbert, 2011).
GP1,2 not only mediates cell attachment and entry but also contributes to immune
suppression by triggering a cytokine storm. This storm, characterised by the
excessive release of pro-inflammatory cytokines, leads to tissue damage,
inflammation, immune dysfunction, vascular leakage, and multi-organ failure
(Wauquier et al., 2010; Volchkov et al., 1998).
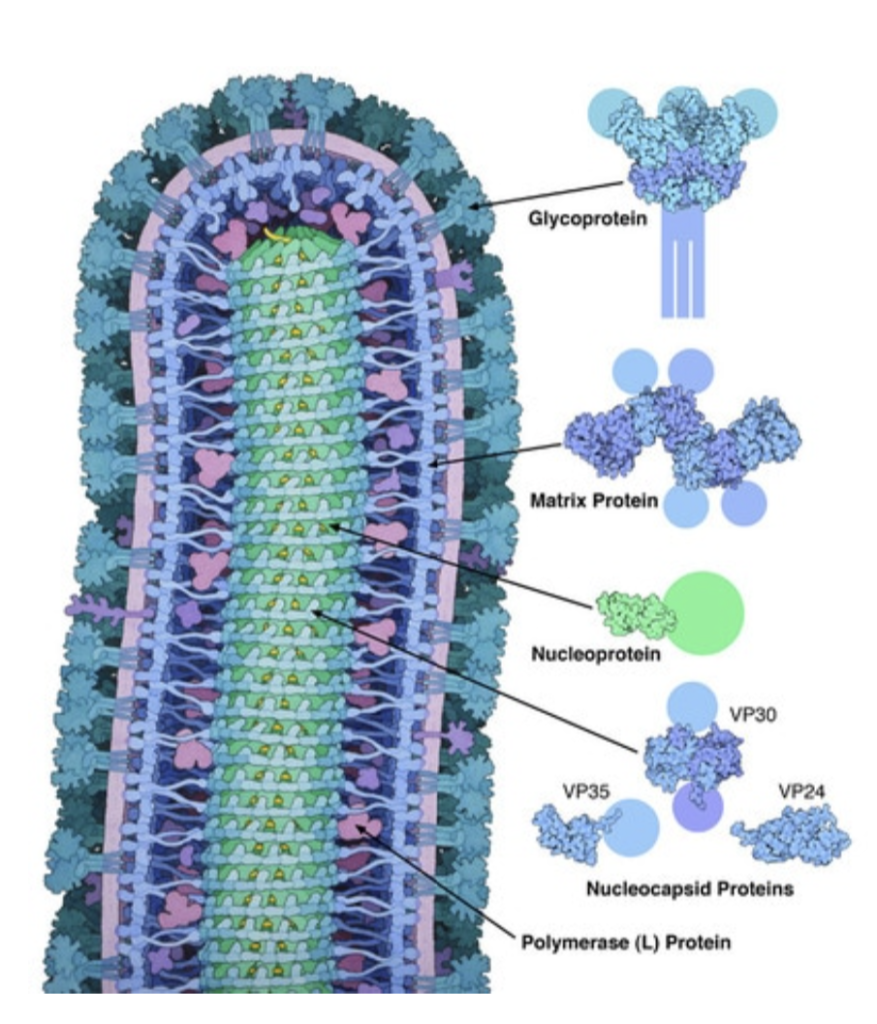
Figure 3: Labelled diagram of EVD in detail.
Ebola’s replication and ability to evade the immune system are further enhanced by
its viral proteins. VP35, for instance, inhibits interferon signalling, a critical
component of the host’s antiviral defences, allowing the virus to spread unchecked
(Basler et al., 2003).
One of the most important questions I had while researching Ebola was ‘what makes
Ebola different and more dangerous compared to other viruses?’ Well, firstly, Ebola
actively targets and destroys key immune cells. By terminating macrophages and
dendritic cells, Ebola suppresses the immune response early in infection, paving the
way for rapid viral spreading throughout the host (Bray & Geisbert, 2005).
Ebola’s GP1,2 protein specifically targets endothelial cells, which line blood vessels.
This leads to the breakdown of blood vessel walls and causes leakage, resulting in
haemorrhaging (Yang et al., 2000). This level of vascular damage and bleeding is
particularly severe in Ebola infections and while other viruses may cause vascular
damage, it’s not to the same extent and often not as systematically.
Ebola also hides in certain parts of the body like the eyes, testes, and brain, where
the immune system has a harder time detecting it, allowing it to persist and
potentially re-emerge later. This has contributed to post-recovery complications
(Sissoko et al., 2017). Many other viruses don’t exhibit this level of persistence after
the main infection clears.

