BY IBRAHEEM MANDA
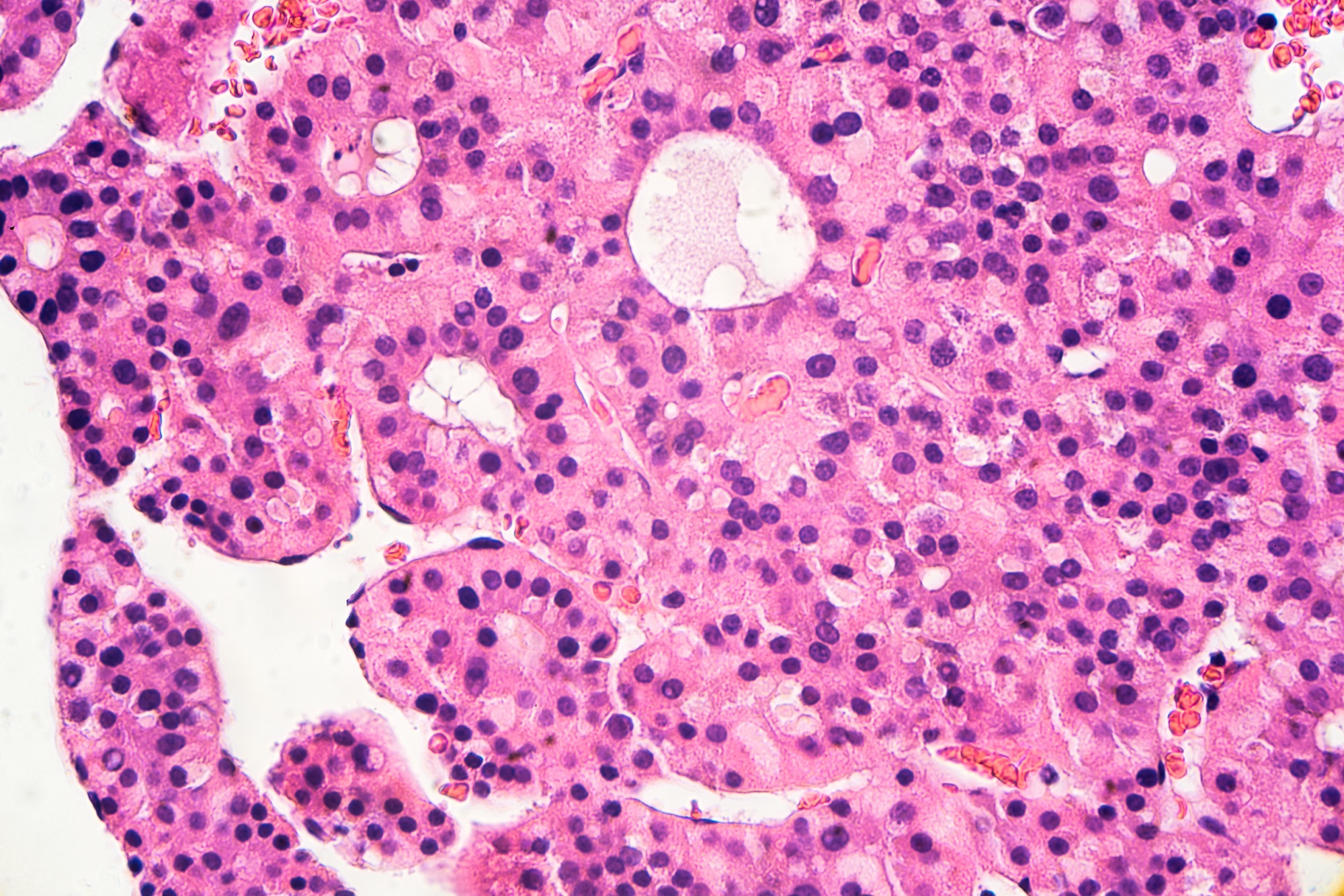
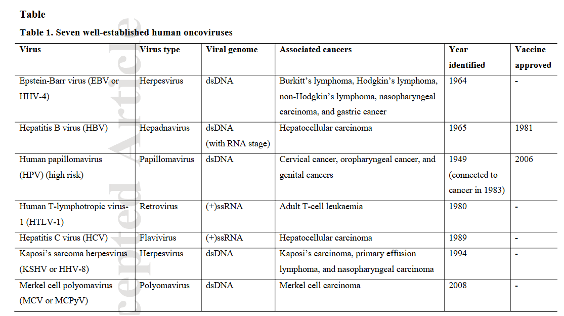
Cancer, a disease often thought of as non-infectious, can surprisingly be linked to viruses that effectively make it ‘contagious’ under specific circumstances. These viruses, known as oncoviruses or oncogenic viruses, are capable of transforming normal healthy cells into malignant ones, leading to cancer. Despite there being numerous viruses, only about seven known oncoviruses are responsible for approximately 15% of cancer cases worldwide.
Oncogenic viruses contribute to cancer development by disrupting normal cellular regulation. They achieve this through a variety of mechanisms, including integrating their viral DNA into the host genome, promoting chronic inflammation, and driving uncontrolled cell proliferation. These disruptions can lead to abnormal cell growth and transformation into cancerous cells.
- Historical background:
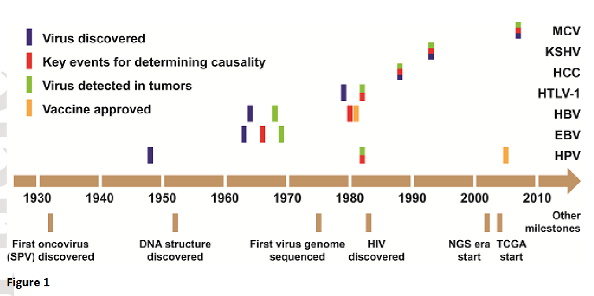
The discovery of oncoviruses has evolved over the years through various scientific approaches. The first oncovirus, the Rous sarcoma virus (RSV), was discovered in 1911 by American virologist Peyton Rous. Rous identified this retrovirus as a cancer-causing agent in chickens. He took a tumor sample from a chicken with sarcoma, ground it up, and filtered the extract to remove any cells, leaving only tiny particles, including viruses. When he injected this filtered extract into healthy chickens, they developed similar tumors. Despite his groundbreaking findings, Rous’s work was initially met with skepticism, as many scientists at the time found it hard to believe that a virus could cause cancer. However, his discovery was eventually recognised, and he was awarded the Nobel Prize in Physiology of Medicine in 1966.
2. Mechanisms of Oncogenesis:
Rous’ discovery in 1911 sparked the idea that viruses could drive tumour formation, since then research has revealed that viruses do not just insert themselves into the host but strategically manipulate the cell’s inner workings. One of the key proteins that many oncoviruses target (HPV, EBV, HBV, HTLV-1) is the p53 protein.
Host integration and Tumour suppression
The virus first enters the body through bodily fluids, open wounds or the sinuses. Some infect specific cells such as epithelial for Human Papillomavirus (HPV) or hepatocytes for Hepatitis B Virus (HBV). Out of the seven known oncoviruses, four integrate their RNA into the host genome (DNA) using reverse transcriptase, turning their viral RNA into DNA (HPV, HBV, MCV, HTLV-1), while one semi-integrates in viral DNA in the form of an episome in the host cell’s nucleus.
After integration, the host cells begin producing viral proteins such as E6 and E7. These proteins degrade the tumor suppressors p53 and Rb (Retinoblastoma protein), which play critical roles in preventing cancer. p53 monitors DNA damage and can initiate apoptosis, while Rb suppresses uncontrolled cell growth by promoting cellular senescence
The virus has now successfully disabled the cell’s biggest safeguards against cancerous growth. Without p53 or Rb, cellular DNA is no longer repaired, mutated cells are not killed by programmed cell death and this spirals into a malignant transformation, a cell that divides uncontrollably, evades the immune system and eventually forms a tumour.
3.Non-integrating oncoviruses and Chronic effects:
EBV, HCV and KSHV are the three oncoviruses that cause cancer without integrating themselves into the host genome but rather by taking advantage of the immune system’s vulnerabilities and creating environments that sustain cancerous cell development.
HCV causes chronic liver inflammation and repeated cycles of cell damage and regeneration that lead to mutations. Reactive Oxygen Species that are generated during inflammation also damage DNA. The combination of these leads to cirrhosis and eventually leads on to hepatocellular carcinoma (primary liver cancer).
EBV is unique in that it is the most advanced virus in terms of immune evasion. By using Latency proteins such as EBNA-1 it avoids MHC presentation which slows detection by T-cells and hence an immune response. Another latency protein LMP1 mimics growth receptors and promotes proliferation. As these infected cells survive and reproduce their unchecked growth leads to lymphomas and carcinomas.
KSHV causes cancer by promoting cell survival and avoiding immune detection through the expression of key viral proteins. It produces LANA, which aids the virus to hide in cells and interferes with tumour suppressor proteins, and vFLIP, which keeps infected cells alive by inhibiting cell death. KSHV also releases vIL-6, a mimic of a human growth signal that encourages inflammation and blood vessel formation. These combined effects allow infected cells to survive, multiply, and eventually develop into cancers such as Kaposi’s sarcoma and primary effusion lymphoma.
4. Epidemiology and Prevention:
Oncoviruses contribute to approximately 15–20% of cancers worldwide, with a disproportionate impact in low- and middle-income countries. HPV is responsible for nearly all cases of cervical cancer, while HBV and HCV are major causes of hepatocellular carcinoma. In regions with high HIV prevalence, KSHV is also a significant contributor to cancer incidence, particularly in the form of Kaposi’s sarcoma.
Several virus-associated cancers are preventable through vaccination and early detection. Vaccines against HPV and HBV have proven effective in reducing infection rates and lowering the incidence of associated cancers. However, access to these vaccines remains limited in many parts of the world. HCV does not currently have a vaccine, though effective antiviral treatments are available. Broader implementation of public health strategies, including vaccination programs, screening initiatives, and access to treatment, is essential for reducing the global burden of virus-induced cancers.
5. The Future:
Research into oncoviruses continues to explore how viral proteins interact with host cell pathways, particularly those involved in cell division, immune evasion, and DNA repair. Techniques such as viral vectors, cell culture models, and structural prediction tools like AlphaFold are used to study these mechanisms. Therapeutic vaccines are in development for viruses such as HPV, and immunotherapies, including checkpoint inhibitors and CAR-T cells, are being investigated for virus-associated cancers. Advances in diagnostics and sequencing are also improving the detection and monitoring of oncogenic viruses, with the aim of supporting earlier intervention and more targeted treatment.
6. Conclusion:
Oncoviruses represent a distinct intersection between cancer biology and infectious disease, contributing to a significant proportion of cancers worldwide. While their mechanisms of oncogenesis vary, many of the cancers they cause are preventable through vaccination, early detection, and public health intervention. As research continues to advance, particularly in the fields of immunology and molecular virology, a clearer understanding of these viruses may lead to more effective treatments and prevention strategies. Their study continues to reveal the complex ways in which infections can influence long-term human health.