Brain tumors are among the most challenging conditions to diagnose and treat effectively. Traditional diagnostic methods such as tissue biopsies, lab-based genomic sequencing, and histopathological analysis are employed to identify the type of tumor, but they often take several days, or even weeks, to yield conclusive results. In critical cases where every hour counts, such delays can limit treatment options and jeopardize prognosis and patient outcomes. But thanks to the rise of Nanopore sequencing technology, this timeframe can be reduced significantly from days and weeks to even hours.
A Diagnostic Breakthrough
In a recent clinical breakthrough, researchers demonstrated that brain tumor types could be accurately identified within 24 hours using real-time DNA sequencing powered by Nanopore technology. In some cases, diagnoses were achieved in as little as one hour, without compromising accuracy compared to traditional methods. The cost of the process and success rate were both on par with current genetic testing methods.
This leap in diagnostic speed is not just an incremental improvement—it’s a paradigm shift. Surgeons could soon receive molecular-level information during or shortly after surgery, enabling more informed decisions about whether to remove more tissue, alter surgical plans based on real- time data, initiate specific therapies, administering targeted drug therapies, or recruiting patients into relevant clinical trials.
What Is Nanopore Technology?
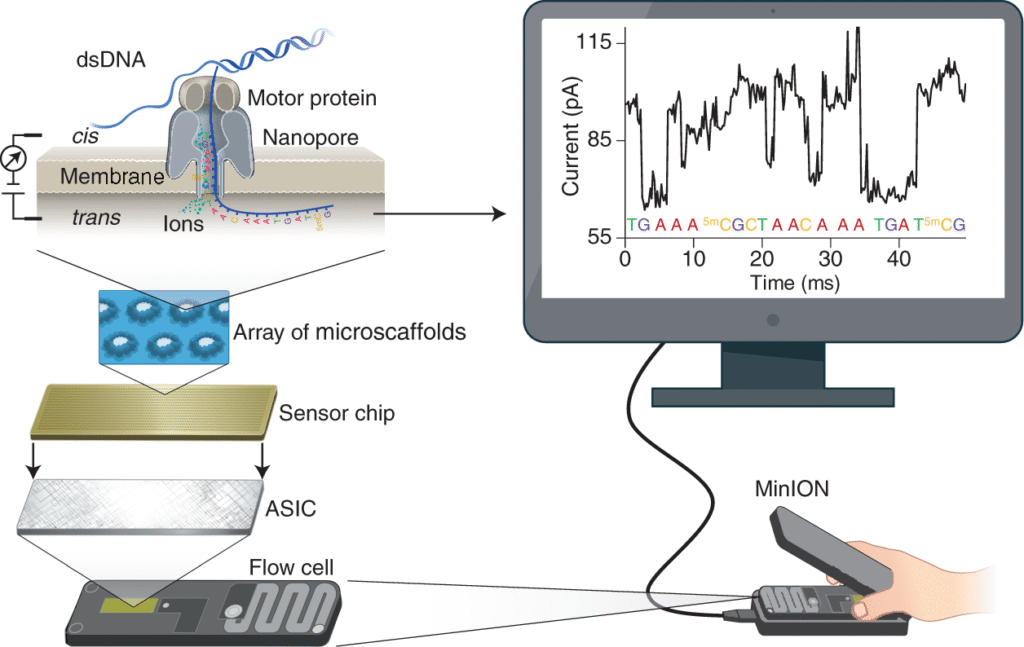
At the heart of this breakthrough lies Nanopore sequencing, a next-generation method for reading DNA and RNA molecules. Unlike conventional sequencing techniques that require amplification and complex sample preparation, Nanopore sequencing works by passing a DNA strand through a microscopic pore embedded in a membrane. An electric current is passed through each of these microscopic pores also called nanopores. As the strand travels through the pore, each base (A, T, C, or G) disrupts the electrical current in a unique way. These changes in the signal are recorded and these current changes are read in real time and translated into genetic sequences using advanced algorithms. This helps in the identification of the order and modifications of DNA bases. The software compares these sequences against known sequences of brain tumours from the gene library thus helping in the diagnosis.
Why use Nanopore Technology?
Nanopore technology can help with on-the-spot diagnosis in operating rooms, ambulances and even in remote places. There are certain key features to this technology that could transform pathology and help surgeons make immediate life-saving decisions.
- Fast – sequencing begins as soon as the DNA starts moving through the pore.
- Portable – devices like the Oxford Nanopore MinION are compact and field-deployable.
- Flexible – capable of reading very long DNA strands, which aids in identifying structural mutations.
Application in Brain Tumors
Brain tumors are particularly complex and diverse at the genetic level. Precise classification is essential for determining prognosis and selecting targeted treatments. The new approach, recently trialed by researchers in the UK, leverages machine learning in conjunction with Nanopore sequencing to match tumor DNA profiles against known genomic patterns of various brain tumor subtypes.
In a recent study, the method correctly identified 90% of brain tumor samples within 24 hours, and 76% within just one hour. The classification accuracy was comparable to standard diagnostic procedures but delivered exponentially faster.
This kind of rapid turnaround could revolutionize:
- Intraoperative decision-making, where surgeons may choose to extend or limit resection based on tumor type.
- Early treatment planning, allowing oncologists to start targeted therapies without waiting for weeks for lab results.
- Remote or underserved settings, where conventional diagnostic labs may not be accessible.
Technical and Practical Challenges
Despite its promise, the widespread clinical adoption of Nanopore technology still faces a few hurdles:
- Data interpretation requires robust machine learning models trained on high-quality tumor databases. Different sample types may require optimisation to prevent bias
- Device standardization and stability under varied hospital conditions must be ensured. The performances of these devices can be affected by temperature, humidity and contamination.
- Regulatory approvals and integration into existing healthcare workflows need careful implementation.
However, the rate of progress is impressive. As technology improves and costs decrease, Nanopore sequencing is becoming an increasingly viable tool for real-time diagnostics.
The Future of Precision Oncology
Beyond brain tumors, Nanopore-based diagnostics are being explored for other cancers, infectious diseases, prenatal testing, and even microbiome profiling. Its real-time capabilities make it especially suitable for precision medicine, where speed and specificity are paramount.
In the context of brain cancer, where survival rates often hinge on timely interventions, this technology could be a game-changer.
Conclusion
The integration of Nanopore sequencing into neurosurgical and oncological practice heralds a new era in ultra-fast, data-driven medical diagnostics. What once took days or weeks can now happen within the same clinical shift, empowering doctors to act faster and smarter in the fight against brain cancer.
As clinical trials continue and the technology matures, it’s not a matter of if but when this innovation will become a standard tool in hospitals worldwide.