BY AUSTIN LAM
Depression as a mental disorder is amongst the most prominent causes of disability, suffered by an estimated 5% of adults, present in approximately 280 million people[1] . Major depressive disorder itself has caused the United States to suffer a societal economic burden totalling at around 333.7 billion dollars extending beyond the primary cost driver of healthcare costs[2]. Identification of depression through a clinical interview has always remained a priority involving analysis of medical history, social history, family history, substance use history coupled with symptomatology[3] . However, current research adopts a new perspective through the lens of genetic and biomarker analysis with emphasis on predicting a patient’s vulnerability to depression. Specifically, decreases in levels of brain-derived neurotrophic factor (BDNF) opposed to alterations in terms of inflammation such as a rise in pro-inflammatory cytokine concentration.
A plethora of existing hypothesis attempt to uncover the pathogenesis of depression ranging from the monoamine theory of depression revising itself around depression recovery[4] , the hypothalamic-pituitary-adrenal (HPA) axis dysfunction hypothesis, the inflammatory hypothesis, the neurotrophic hypothesis, and others exploring genetic and psychosocial aspects[5].
One may ask why its pathophysiology has not been summarized through a unified theory. The answer is that all respective theories are only applicable to certain pools of patients, exemplified through the heterogeneity of major depressive disorder. This provides an explanation of the holistic approach clinicians favour involving a combination of psychological and pharmacological approaches[6] . Indeed, antidepressant treatment has its limitations, revealed through a lack of response towards typical antidepressant medication in 10-30% of patients suffering from major depression[7].
In this way, the possibility of utilizing BDNF and inflammation levels as a method of differentiating between healthy and depressed patients will provide further insight into both the etiology and mechanism of depression, investigating the role of inflammation and BDNF along with their interplay assessed in context.
Brain Derived Neurotrophic Factor
The neurotrophin brain-derived neurotrophic factor (BDNF) is a member of the neurotrophin family. Initially purified from mammalian brain, BDNF was the first isolated neurotrophic factor since nerve growth factor NGF [8] , crucial for neuronal growth, survival, differentiation and neural plasticity, not solely during development but even in adulthood[9] . Being involved in brain disorders such as depression, PTSD and many others, its role within existing antidepressants remains extensively studied[10]. Encoded by the BDNF gene, its expression in humans is controlled by an antisense transcript BDNF-AS forming RNA duplexes to modulate translation[11]. The BDNF gene consists of 11 exons with 9 functional promoters; exon IX encodes BDNF protein, whereas various mRNA isoforms are controlled by remaining exons. In the brain, BDNF is widely distributed in the CNS, especially prevalent within the cerebral cortex and hippocampus[12] where they would contribute to memory, learning and long-term potentiation[13]. BDNF acts upon neurons[14], promoting activation of astrocytes and microglia[15] . However, its presence within the heart, spleen, skin tissue[16] , circulating in the bloodstream mainly through blood platelets[17] where preparation of peripheral BDNF analysis can be performed through preparation via serum, plasma and platelets[18], indicating the non-neuronal existence of the protein.
BDNF is first synthesized within the endoplasmic reticulum (ER) as its precursor protein (preproBDNF), later translocated to the Golgi apparatus, as signal sequence of the pre-domain is cleaved forming proBDNF. In order to achieve its mature isoform (mBDNF), proBDNF can be proteolytically cleaved intracellularly by enzymes such as furin or protein convertases[19], or extracellularly by proteases[20]. It is important to note how proBDNF and mBDNF preferentially bind to different receptors exhibiting opposing results clearing the common misconception of proBDNF as an inactive precursor. ProBDNF bind to the p75NTR receptor inducing neuronal apoptosis[21] , whilst mBDNF preferentially binds to tyrosine kinase receptor tropomycin receptor kinase B (TrkB).
TrkB activation by BDNF with high affinity activates downstream responses, inducing dimerization and autophosphorylation of intracellular tyrosine residues, acting as docking site activating signalling pathways: Ras/MAPK-ERK, PI3-K and PLCγ pathway (fig. 1). Eventually transcription factor CREB is activated, responsible for gene expression regarding molecules required for neuronal differentiation and survival.
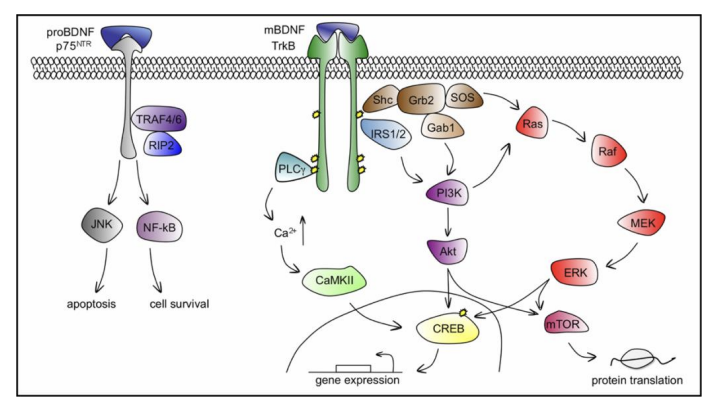
Figure 1 – depicting the BDNF–TrkB and BDNF–p75NTR signalling pathways. Image source: Cunha (2010). A simple role for BDNF in learning and memory? Frontiers in Molecular Neuroscience. doi:https://doi.org/10.3389/neuro.02.001.2010.
Alterations in BDNF expression would be due to changes in its transcript regulation contributed a multitude of different factors. The findings of a study conducted by (Lubin et al., 2008) exemplify modulation of BDNF expression through epigenetic mechanisms via contextual fear learning, considering DNA methylation alone can oversee exon-specific BDNF gene transcription. This confirms that environmental impact is sufficient to induce changes in BDNF expression[22] . Chronic stress reduces BDNF levels typically in the hippocampus[23] . Multiple lines of evidence prove aging to result in a decrease in plasma and serum levels of BDNF[24,25] , though others concluded there was no association between age and BDNF levels with the lack of any change[26,27]. Factors such as exercise [28] promote expression whereas its levels are found to be decreased throughout many neurogenerative diseases such as Parkinson’s disease[29] and Huntington’s disease[30] .

